Your everyday world from a physicist's perspective
Time for tea
A little experiment to make it through the cold winter days:
- Boil some water to make yourself some strong English tea (no milk please);
- Take a drinking glass and fill it to the top with cold water from the tap;
- Use a spoon to gently put some of the hot tea on top of the cold water;
- Sit back and see what happens (you might want to put a white sheet of paper behind the drinking glass to have more contrast)
What exactly do we see?
To understand this phenomenon I first have to inform you on two facts of nature: (1) water plus tea is heaver than water without tea and (2) water of 100 °C is less dense than water of 15 °C.
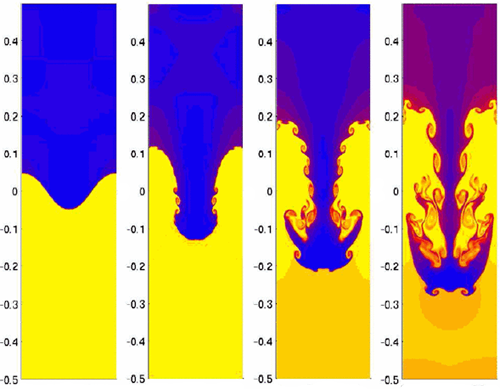
Initially, the second effect is dominant, and the hot tea will float comfortably on top of the cold water. But as the tea cools down, it must become heavier than the water below it at some point! In theory, a light fluid can support a denser fluid just fine. For example, in theory the air pressure at sea level is high enough to hold a water column of about 10 meters up against the ceiling. But if someone ever flipped a full bucket of water above your head, you know that this is not true in practice! This also holds for our tea on water system, although here the effect is somewhat more subtle.
The tea-water interface starts out more or less flat, but when the tea becomes heavier than the water below, this situation does not represent a stable equilibrium. By an analysis very similar to the one in the article on tumbling books, one can show that small perturbations of the interface are amplified and grow quickly over time (exponentially). This instability manifest itself as a periodic array (look at your glass from the top) of so called 'Rayleigh-Taylor fingers' that whirl down with a kind of mushroom shaped heads. Figure 2 below shows a beautiful numerical simulation of such a plume.